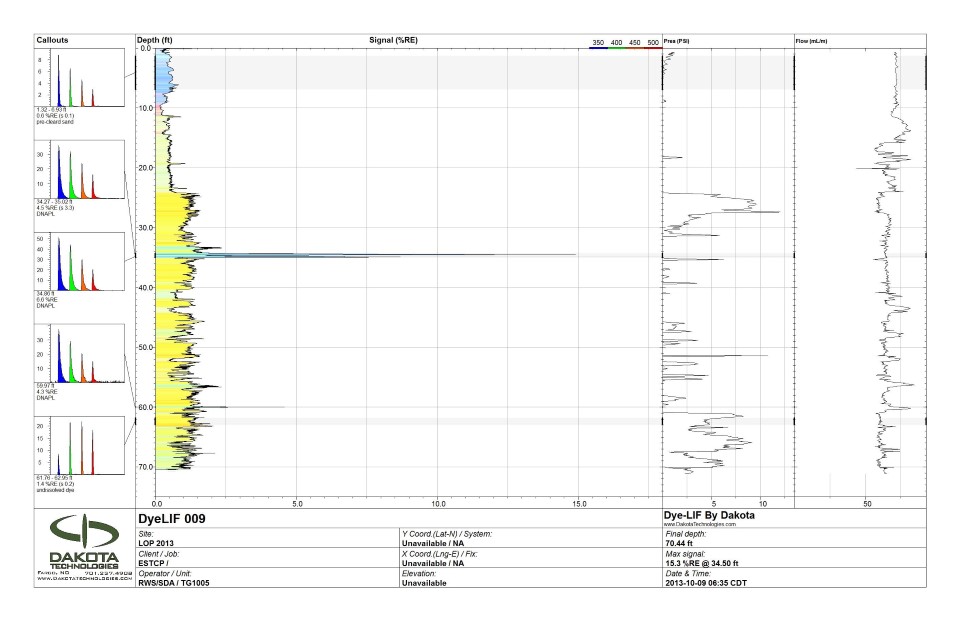
The capability of the DyeLIF was recently demonstrated at a small footprint chlorinated spill site in Massachusetts. Tests of chlorinated solvent DNAPL recovered from the site revealed a mixture of 59% trichloroethylene, 34% 1,1,1-trichloroethane with various solvents and petroleum hydrocarbons making up the balance. Local geology in the demonstration area can be generally classified as fine sand and silt with intermittent silt and clay layers. A continuous silt layer is observed at 14 m below ground surface (bgs), which is consistent with other areas of the larger site.
The log image below illustrates one of the raw DyeLIF logs obtained. The light blue color of the upper 3m (10 ft) fluorescence is from loose sand that was placed in the hole after pre-clearing for utilities. Notice the unique wave-form in callout one, showing the waveform from the sand. The yellow-filled baseline response is the weak fluorescence emitted by un-solvated dye particles injected into the formation. The other more intense blue-filled spikes at various depths are narrow responses of DNAPL. Notice the longer lifetimes and blue-shifted waveforms in callouts two, three, and four. This waveform shape is unique to DNAPL-solvated dye, establishing with high confidence the origin of the fluorescence response at those depths. The bottom callout shows the red-shifted waveform with very short lifetimes. The log is typical of logs in which modest DNAPL detections were made, but the pore saturation was low enough that the DNAPL response is obscured to some degree by un-solvated dye and other background fluorescence.
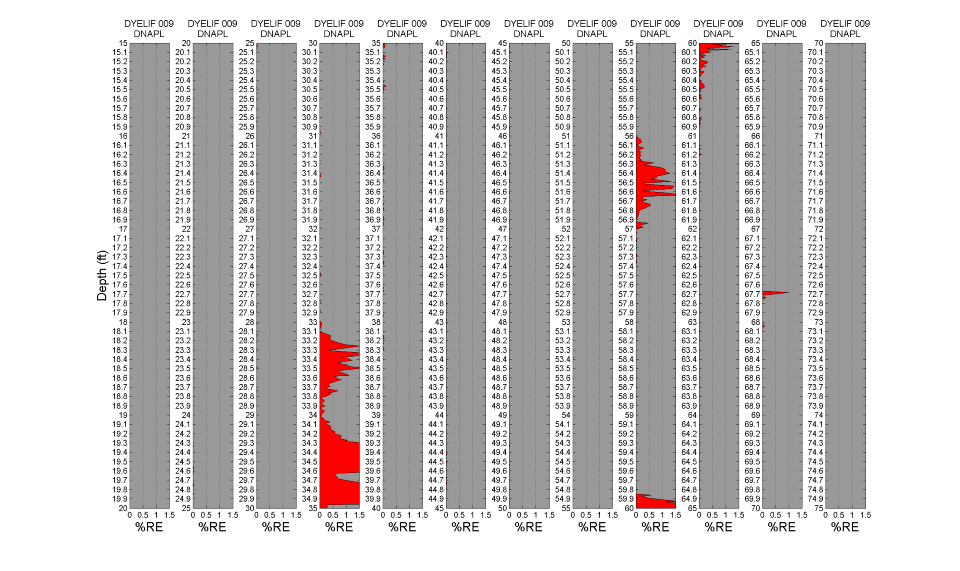
LIF waveforms are stored with each and every depth analyzed, allowing for post-processing using a non-negative least squares (NNLS) fitting process. The process uses a basis set of waveforms (non-solvated dye, DNAPL-solvated dye, sand, etc.) to determine the origins of and then appropriate the fluorescence response. As discussed, there are dramatic spectral and temporal differences in the waveforms for DNAPL solvated dye vs. un-solvated dye. This allows us to mathematically strip away all but the DNAPL-specific responses, yielding high-confidence DNAPL-specific logs of DNAPL detections. The image below shows the same log after such post-processing. So that even small (but critical) DNAPL ganglia are readily recognized, the NNLS data is plotted in many columns, each five feet (1.5 m) in height. The fluorescence scaling of the processed log is held to 0-5 %RE so that even the lowest of pore saturations are obvious. The tiny response at 67.7 ft bgs is an example detection of a very small DNAPL ganglion. This post-processing requires about 5 minutes to conduct and was done on site immediately after the DyeLIF log was collected. Note the data in this plot is zoomed in such that each column represents 5 feet of depth.